INTRODUCTION
Infraorbital nerve (ION) dysfunction is commonly reported after zygomaticomaxillary complex (ZMC) fractures (in 30.4%–80% of cases), as these fractures frequently involve the infraorbital fissure, canal, or foramen [
1-
5].
ION dysfunction may not recover spontaneously in some patients with severe ZMC fractures; however, spontaneous recovery of the nerve does occur in most cases [
6]. However, little data are available concerning sensory change in the areas (lower eyelid, cheek, lateral wall of nose, lip and gingiva/teeth) innervated by the ION. In our experience, at 3–4 months following ZMC fractures, patients often complain about impaired sensory function in the lip despite full recovery in other areas. Therefore, there are differences in sensory recovery among the areas in which the ION is distributed. Homer et al. [
7] assessed sensory changes in the areas along the ION path, but they used only a visual analog scale (VAS) for sensory assessment. However, a limitation of VAS assessments is that they rely on patients’ subjective judgment. Therefore, we evaluated sensory changes in four designated areas innervated by the ION using the static two-point discrimination test (TPD) and the vibration perception threshold test.
Trigeminal sensory afferent fibers mediate the blink reflex, as well as their central connections in the trigeminal and facial brainstem nuclei at the level of the lower pons and medulla oblongata [
8]. When the skin innervated by the ION is electrically stimulated, the contraction response of the orbicularis oculi muscle via the pontine reflex or pontine and lateral medulla pathway is electromyographically evaluated. Therefore, changes in the compound muscle action potential (CMAP) can be observed when ION sensory afferent fibers are injured. Ohki and Takeuchi [
9] reported that the blink reflex is an objective method for evaluating ION injuries, unlike other evaluation methods that depend on patients’ verbal responses. For this reason, the blink reflex may be useful in evaluating ION injuries following ZMC fractures. Therefore, our aim was to evaluate the diagnostic significance of the blink reflex in patients with ION dysfunction after ZMC fractures.
METHODS
Patients
Subjects with unilateral ZMC fractures who were treated with open reduction and internal fixation with or without orbital floor reconstruction between December 2015 and March 2019 participated in this study. All participants complained of some degree of ION dysfunction preoperatively. This study was approved by the Institutional Review Board of Sungkyunkwan University (IRB No. 2020-06-004). The inclusion criteria encompassed patients who were at least 18 years old and had received at least 4 months of follow-up. The exclusion criteria were patients who had a facial skin injury or associated maxillofacial fractures such as nasal bone fracture, a history or presence of facial nerve palsy, or pregnancy.
Procedure
All reduction surgery was performed under general anesthesia. All procedures were performed using a lateral eyebrow, subciliary, and intraoral approach and three-point fixation of the zygomaticofrontal suture, infraorbital rim, and zygomaticomaxillary buttress. Titanium plates were used for rigid fixation. The orbital floor was routinely explored and, if necessary, reconstructed using a Medpor porous polyethylene implant (Porex Surgical Inc., Newnan, GA, USA) to restore orbital volume.
Assessment
All assessments were performed by a physiatrist (YSP) preoperatively (T0), 1 month postoperatively (T1), and at least 4 months postoperatively (T4). The initial assessment was performed on the day before the operation to measure facial swelling reduction as best as possible. The TPD and vibration test were assessed in four designated areas along the ION tract: lower eyelid, cheek, lateral wall of nose, and lip. The gingiva was not tested to prevent the need for lifting the lip, which would affect the assessment and cause patient discomfort. The testing conditions were identical for all subjects.
Patients’ subjective sensory function was assessed using a VAS (range, 0–10). A score of 0 denoted normal intensity sensation and 10 represented no sensation.
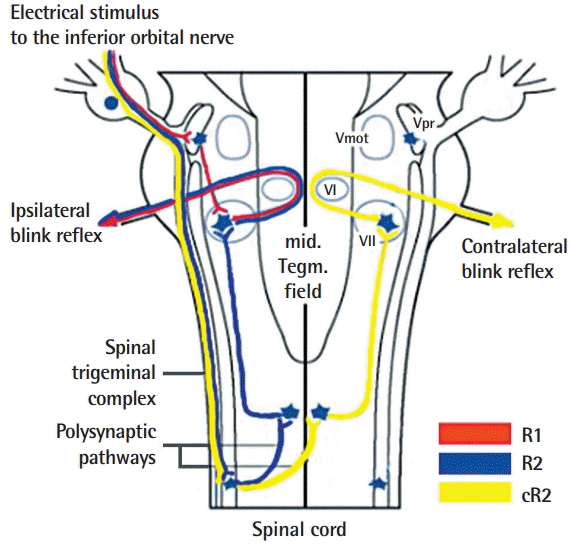
In the infraorbital blink reflex, the eyelid closes in response to an exteroceptive/nociceptive stimulus to the ION. Patients were asked to close their eyes while in supine position. Active electrodes were placed bilaterally over the orbicularis oculi muscle at the lower eyelids, and a reference electrode was placed on the bilateral temples, lateral to the eye. Stimulating electrodes were pasted on the skin just above the infraorbital foramen. A ground electrode was placed on either the chin or forehead. The stimulation intensity was 20–25 mA with a duration of 0.1 ms at a frequency of 0.5 Hz. Nicolet Viking Select (Biomedical, Viasys Neurocare, Madison, WI, USA) was used with settings of sensitivity of 200 μV/division, a sweep speed of 10 ms/division, and a filter at 20–200 Hz. The three recorded responses consisted of an ipsilateral early component (R1) followed by ipsilateral and contralateral late components (R2 and cR2, respectively) (
Figs. 1,
2). Responses were defined as acceptable, prolonged (ipsilateral R1 ≥13.1 ms, ipsilateral R2 ≥41 ms, or contralateral R2 ≥44 ms), or no response. An acceptable response was assigned a value of 3, a prolonged response was assigned a value of 2, and no response was assigned a value of 1 [
10].
To measure the TPD, a Dellon-McKinnon Disk-Criminator (Sammons-Preston Canadian Inc., Mississauga, ON, Canada) was used. The measures were transformed into categorical data as follows: a TPD of 1–5 mm was given a value of 4, a TPD of 6–10 mm was given a value of 3, a TPD of 11–15 mm was given a value of 2, and an unmeasurable TPD was given a value of 1 [
11].
To measure the vibration perception threshold, an MMD-1000A Biothesiometer (Medical Supply Co., Seoul, Korea) was used. The measurements were transformed into categorical data as follows: a vibration perception threshold up to 15 V was assigned a value of 3, a vibration perception threshold of 16–25 V was assigned a value of 2, and a vibration perception threshold of >25 V was assigned a value of 1 [
12].
Statistical analysis
The results are presented as mean values. The analysis was performed using a linear mixed effects regression model. All models included the following covariates: sex, age, smoking status, orbital floor reconstruction requirement, and time. If time was significant, Bonferroni-adjusted pairwise comparisons were performed to determine significant changes between periods (T0 and T1, T0 and T4, and T1 and T4). Their respective differences and 95% confidence intervals (CIs) were calculated, and P-values of 0.05 or less were considered to indicate statistical significance. All statistical analyses were performed using Stata version 14.0 (StataCorp, College Station, TX, USA).
DISCUSSION
In the infraorbital blink reflex, the afferent portion of the blink reflex is supplied by the ION, and the efferent volleys are conducted by branches of the facial nerve that innervate the orbicularis oculi muscle. When the skin innervated by the ION is electrically stimulated, a CMAP from the surface of the orbicularis oculi muscle is recorded. When the ION is injured, a change in the CMAP can be observed. ZMC fractures are commonly accompanied by ION dysfunction. Therefore, in patients with ZMC fractures, the presence of an abnormal infraorbital blink reflex may indicate nerve injury along the ION-facial nerve pathway.
The CMAP is normally composed of three responses: R1, R2, and cR2. The R1 response is mediated by an oligosynaptic pontine circuit. The R2 and cR2 components result from activation of a polysynaptic chain of brain stem interneurons extending in the lateral reticular formation at the pontine and medullar level [
8,
13]. Because the circuit of R1 is shorter than that of R2 and cR2, R1 is recorded earlier than R2 and cR2. The efferent volley of the R2 response involves the ipsilateral facial nerve, but that of the cR2 involves the contralateral facial nerve. Therefore, when the ION is injured, R1, R2, and cR2 may show similar changes because the afferent portions are in the ION. In our study, six patients with an abnormal infraorbital blink reflex had identical grades of R1, R2, and cR2. However, one patient had prolonged R1 and acceptable R2 and cR2. Recently, Ohki and Takeuchi [
9] noted that R1 and R2 may be mediated by different sensory nerve fibers: R1 by A-β fibers and R2 by A-β and A-δ fibers. The thickest type of fiber is A-α, followed in descending order by A-β, A-δ, and C. Thicker fibers are more susceptible to compression or inflammation [
14]; thus, the fibers related to R1 may be more fragile [
9]. Therefore, we expected abnormal R1 to be more sensitive to ION injuries. However, we did not observe a significant difference between R1, R2, and cR2.
Preoperatively, on the VAS, three patients had normal sensation, and 15 patients had paresthesia. Seven patients had an abnormal infraorbital blink reflex, indicating nerve damage more severe than neurapraxia, as patients with an abnormal blink reflex might have more severe symptoms than patients with a normal blink reflex. Therefore, although 15 patients complained of some degree of ION dysfunction, only seven (46%) of these patients had nerve damage identified in the infraorbital blink study. Recovery of the VAS scores required less time than that of the infraorbital blink reflex. Compared to the preoperative results, the VAS significantly improved at 1 month postoperatively, while the infraorbital blink reflex significantly improved by the final follow-up.
In our experience, many patients complained about affected sensory perception in the lip despite normal sensation in other areas. Nerve recovery is better after distal injuries than after proximal ones [
15]. The lip is the farthest distance from inferior orbital canal or foramen. These structures are commonly damaged by ZMC fractures that disrupt the ION-mediated innervation of target organs. Therefore, we predicted that the lip would show the latest sensory recovery of the studied structures. Our results showed that vibration perception threshold of the lip had not improved by the final follow-up, but TPD of the lip had significantly improved. Gelberman et al. [
16] reported that threshold tests such as the vibration and Semmes-Weinstein monofilament tests consistently better reflected gradual changes in nerve function than innervation density tests like TPD and moving TPD. Therefore, sensory recovery of the lip tends to occur later than that in other areas innervated by the ION.
Age is a factor contributing to peripheral nerve recovery, which is usually slow and partial in patients older than 50 years [
17,
18]. However, in our study, age did not have an influence on recovery from ION injury. This may be a result of the small number of patients included or the fact that only mild ION injuries were analyzed. In our study, most patients had recovered from ION dysfunction within 16 weeks. Therefore, most patients included in our study did not have more severe nerve injury than neuropraxia. When a nerve is injured mildly, as in these cases, nerve recovery may not be influenced by age. We hypothesized that ZMC fractures accompanied by inferior orbital wall fractures would show slower sensory recovery than ZMC fractures alone, since the ION is routed in the floor of the orbit. However, when the TPD and vibration perception threshold were assessed in four designated areas along the ION pathway, only the lip was influenced by inferior orbital wall fracture. Homer at al. reported that patients with isolated floor fractures noted long-term numbness in the cheek only, suggesting that the permanent injury was isolated to a single ION branch [
7,
19]. Therefore, inferior orbital wall fractures have a high probability of causing nonselective injuries of a single ION branch.
Our study has several limitations. First, the sample size of the present study was insufficient to ensure the validity of the findings. Although 109 patients were enrolled in our study, only 18 completed the study. This may result from the long measurement duration of 15 to 20 minutes for blink testing, which can be difficult in long-term follow-up. Some people experienced discomfort from the electrical stimulus. Second, the average follow-up period was 4.7 months; this may not have permitted time for neural regeneration, which may take more than 6 months postinjury [
20]. However, at the last follow-up examination, most patients had recovered from ION damage. Therefore, the assessment of sensory recovery was not an issue. Third, we did not assess the possible association between ION dysfunction and the ZMC fracture pattern, which should be investigated in future studies.
The blink reflex test is an electrophysiological technique that is used to assist in the evaluation of patients with suspected trigeminal or facial nerve disorders or demyelinating polyradiculopathies [
8,
21-
24]. Ohki and Takeuchi [
9] evaluated the objectivity of the infraorbital blink reflex in patients with maxillary lesions and concluded that the infraorbital blink reflex is an objective method for evaluating ION injuries. Therefore, we used the blink reflex as an objective and quantitative method to evaluate ION injuries following ZMC fractures, and we demonstrated that the infraorbital blink reflex reflected the recovery of ION dysfunction.