

Author contribution
Conceptualization: RN Bogdasarian, SB Cai, A Ignatiuk, ES Lee. Data curation: RN Bogdasarian, BNN Tran. Formal analysis: RN Bogdasarian, SB Cai, BNN Tran. Methodology: RN Bogdasarian, SB Cai, A Ignatiuk, ES Lee. Project administration: RN Bogdasarian, SB Cai. Visualization: RN Bogdasarian, SB Cai, BNN Tran. Writing - original draft: RN Bogdasarian. Writing - review & editing: RN Bogdasarian, BNN Tran.
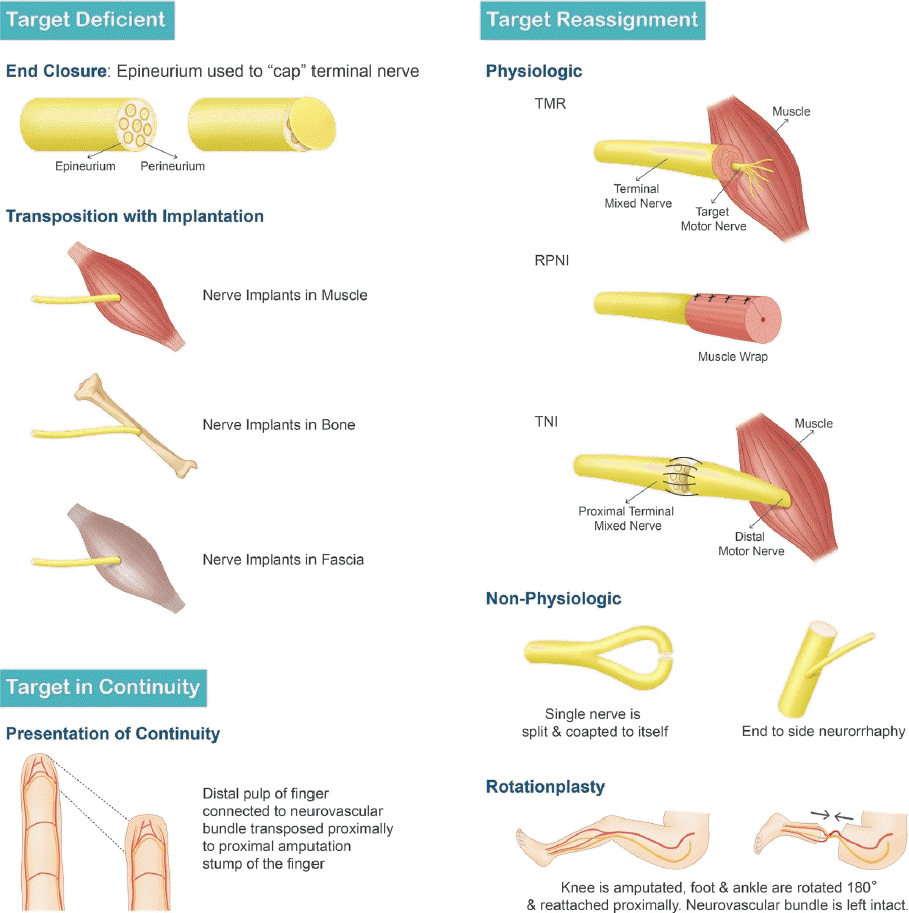
| Author | Category/Technique | Purpose | Design | Sample size | Analysis techniques | Quantitative results | Strengths | Limitations |
|---|---|---|---|---|---|---|---|---|
| Yuksel 1997 [70] | Target deficient/End closure | To compare epineural ligatures, flaps and grafts for the prevention of amputation stump neuromas | Prospective, randomized clinical trial | 23 Patients, 48 nerves | Tinel’s sign used to elicit pain, recorded on a subjective 10-point VAS for pain. ANOVA | Pain scores 0-10: Ligatures 5.18 | Single surgeon, randomized, varied techniques in single patients. > 6 Months follow up | Heterogeneous amputation causes, unclear if blinded, subjective pain scale |
| Flaps 4.25 | ||||||||
| Grafts 2.06 | ||||||||
| Grafts resulted in significantly less neuroma pain P < 0.05 | ||||||||
| Gorkisch 1984 [9] | Target reassignment: non-physiologic/ | To examine the efficacy of centro-central nerve union for the prevention of hand neuromas | Prospective, nonrandomized, nonblinded cohort | 30 Patients | Physical examination, subjective reports | Only 1 of 30 patients returned with clinical neuroma over 4 years follow up. | Prospective | No statistical analysis or raw data presented. Selection bias, nonrandomized, nonblinded, assessor bias |
| Neurorrhaphy | ||||||||
| Belcher 2000 [67] | Target reassignment: non-physiologic/ | To compare direct digital nerve CCU to simple nerve transection (control) in finger amputations | Prospective randomized double-blinded clinical trial | 31 Digits (control: 16, CCU: 15) | Subjective questionnaire. Objective S2PD, Dolorimeter, grip strength. | Subjective sensation better in control than CCU (P < 0.02). Objective tenderness better in CCU than control (P < 0.001). Grip strength equal. | Prospective, randomized, objective measures, double-blinded. 2 Years follow up | Transfer bias (half lost to follow up) |
| Neurorrhaphy | Students t-test, Mann-Whitney test | |||||||
| Economides 2016 [71] | Target reassignment: non-physiologic/ | To propose tibial and common peroneal nerve coaptation at the time of amputation as means to prevent TNPLP | Prospective cohort | Coaptation cohort: 6 | VAS, neuropathic pain medication use, neuroma formation, presence of phantom limb pain, prosthetic tolerance, and ambulatory status; 2-tailed, unpaired t-test and chi square test for data containing continuous and categorical variables | 6 Months: VAS scores (0.75 vs. 5.6; P = 0.02) as well as neuroma (0% vs. 54.5%; P = 0.03) and phantom pain (0% vs. 63.6%; P = 0.01) remained lower among patients who underwent coaptation. At follow-up, 67% of coaptation patients were ambulating with a prosthesis vs. 9% of neurectomy patients (P = 0.01) | Prospective | No disclosure of randomization, blindness, patient demographics comparison or number of surgeons involved |
| Neurorrhaphy | Traction neurectomy control: 11 | |||||||
| Pet 2014 [15] | Target reassignment: physiologic/ | To investigate if TNI prevents neuroma formation in acute traumatic amputation | Retrospective chart review | 12 Patients | Chart review for palpable neuroma pain | 11 of 12 patients were free of palpable neuroma pain | > 8 Months follow up | Retrospective, nonrandomized, subjective pain data, transfer bias, selection bias, assessor bias |
| Targeted nerve implantation | ||||||||
| Souza 2014 [19] | Target reassignment: physiologic/ | To evaluate the effect of TMR on residual neuroma pain | Retrospective chart review | 11 Without neuroma; 15 with established neuroma | Chart review for complete resolution of pain and fit with TMR-controlled prosthesis | None of the 11 patients who presented without neuroma developed a neuroma after TMR | > 6 Months follow up | Retrospective. Neuroma prevention not a primary objective of the study. TMR primarily performed for control of upper extremity prosthetics, not neuroma prevention. No objective pain scale |
| Targeted muscle reinnervation | ||||||||
| Bowen 2017 [6] | Target reassignment: physiologic/ | To show that TMR, alone and in conjunction with other methods, is a reliable treatment for terminal neuroma and phantom limb pain | Literature review and presentation of experience | 20 Patients | Not disclosed | Not disclosed | Well organized, thorough | No disclosure of article selection protocol in primary publication, prevention of neuromas or phantom limb pain was not purpose of review |
| Targeted muscle reinnervation | ||||||||
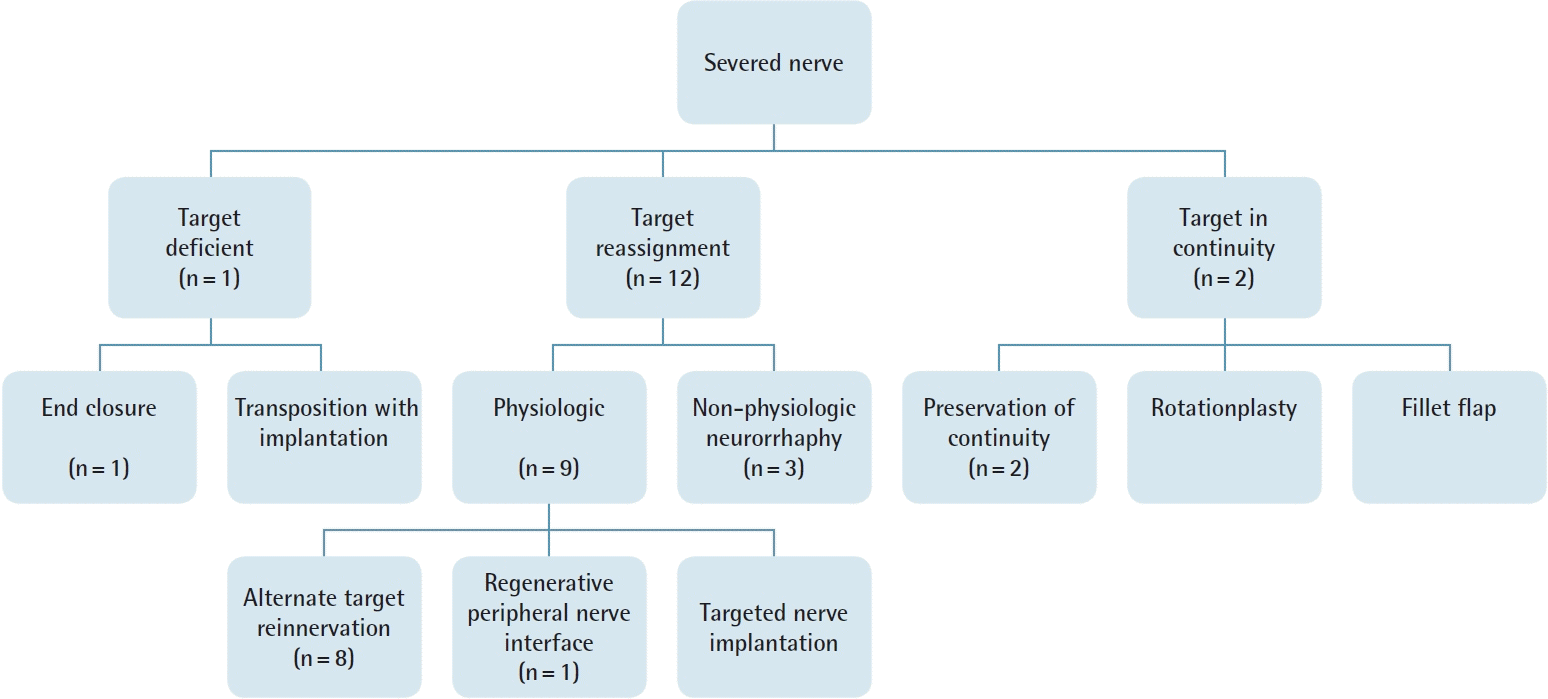
| Ives 2017 [11] | Target reassignment: physiologic/ | To review the current literature on the treatment of terminal neuromas | Comprehensive literature review | 98 Articles cited | 4 Treatment categories formulated with report and interpretation of data | 4 Categories include epineural closure; nerve transposition with implantation; neurorrhaphy, and alternate target reinnervation. Minimal quantitative evidence regarding prevention given | Well organized, thorough review | No disclosure of article selection protocol in primary publication, prevention of neuromas or phantom limb pain was not purpose of review |
| Targeted muscle reinnervation | ||||||||
| Kuiken 2017 [72] | Target reassignment: physiologic/ | To present the technique of TMR in upper and lower extremity amputations | Technique presentation | 100 Patients | Presentation of methods, experience and literature | 1 of 100 patients who underwent TMR were re-explored to resect a neuroma. | Well organized, adhere to strong surgical principles | Neuroma presentation a secondary topic, no statistical analysis, minimal presentation of raw data, assessor bias |
| Targeted muscle reinnervation | ||||||||
| Alexander 2019 [73] | Target reassignment: physiologic/ | To assess TNPLP specifically in patients who undergo oncologic amputation | Single institution cohort study | 27 Patients underwent oncologic amputation at a single institution and were compared to 58 patients treated at other institutions | PROMIS looking at pain intensity, pain behavior, and pain interference | Mean differences in PROMIS scores for TNPLP were 5.855 (P=0.15), 5.896 (P=0.033), 7.435 (P=0.011) for pain intensity, pain behavior and pain interference, respectively. | Prospective, utilization of standardized pain related measures | Nonrandomized, nonblinded, multimodal approach to postoperative pain control could a confounder, of 27 patients who underwent TMR, only 15 had follow up greater than 1 year despite median follow-up of 16 months |
| Targeted muscle reinnervation | Mean differences in PROMIS scores for residual limb pain were 5.477 (P=0.031), 6.195 (P=0.028), 6.816 (P=0.014) for pain intensity, pain behavior and pain interference, respectively. | |||||||
| Bowen 2019 [74] | Target reassignment: physiologic/ | To present results of TMR in BKA as a means of preventing TNPLP | Case series | 22 Patients (18 primary, 4 secondary) | Physical exam, subjective patient reporting | 72% of the primary TMR cohort experience phantom limb pain in the first month, with a decline to 19% at 3 months, and 13% at 6 months. | 1 Year follow up | Nonrandomized, nonblinded, data specific to BKA |
| Targeted muscle reinnervation | ||||||||
| Valerio 2019 [75] | Target reassignment: physiologic/ | To assess if TMR at the time of amputation decreases incidence and severity of TNPLP | Multi-institutional cohort | 51 Patients compared with 438 major limb amputees | 11 NRS and PROMIS looking at pain, intensity, behavior, and interference | TMR showed lower median PROMIS t-scores for TNPLP with pain behavior (P<0.001), pain intensity (P<0.001) and pain interference (P<0.001). A similar pattern was seen with residual pain in regards to pain behavior (P<0.001), pain intensity (P<0.001) and pain interference (P<0.001). | Prospective, utilization of standardized pain related measures | Non-randomized, nonblinded, referrals for amputee group at discretion of surgical team performing amputation, 3-month minimum follow-up time |
| Targeted muscle reinnervation | ||||||||
| Kubiak 2019 [66] | Target reassignment: physiologic/ | To present results of creation of RPNI as a means of preventing TNPLP and neuromas | Retrospective | 45 Patients underwent RPNI matched to 45 control patients selected from a pool of 178 | Documentation of physical exam findings and patient reporting. Fischer exact test | No symptomatic neuromas noted in the intervention group (0% vs. 13.3%, P=0.026). There was a reduced incidence of TNPLP in the intervention group (51.1% vs. 91.1%, P<0.0001). | Long mean duration of follow-up on average of 1 year | Inconsistence chart documentation |
| RPNI | ||||||||
| De Smet 1996 [68] | Target in continuity/ | To present results of mid-finger amputation reconstructions with bi-neurovascular bundle pedicled volar pulp flaps | Case series | 4 Patients | Personal experience and opinion | All 4 flaps survived with sensibility equal to preoperative | May be beneficial when indicated | Nonrandomized, nonblinded, small sample size, no comparison. minimal long-term follow-up, assessor bias |
| Preservation of continuity | ||||||||
| St-Laurent 1996 [69] | Target in continuity/ | To assess results of elective digital amputation with bipedicled neurovascular volar pulp flap | Case series | 8 Patients, 9 amputations | Physical exam, patient reporting | 7 Patients without painful neuroma, 1 patient with preoperative pain that did not improve postoperatively. | Prospective | Minimal long-term follow-up (range, 2–9 months), small sample size |
| Preservation of continuity |
ANOVA, analysis of variance; BKA, below-knee amputation; CCU, centro-central union (the direct union of two nerve endings, or the splitting and union of a single nerve ending); NRS, numerical rating scale; PROMIS, Patient-Reported Outcomes Measurement Information System; RPNI, regenerative peripheral nerve interface (reinnervated, non-vascularized muscle grafts; TMR, targeted muscle reinnervation (the coaptation of mixed nerves into terminal motor nerves for the purpose of creating functional electromyographic signals; TNI, targeted nerve implantation (coaptation of nerves solely for the purpose of neuroma prevention); TNPLP, terminal neuroma or phantom limb pain; S2PD, static two-point discrimination; VAS, visual analog scale.